While BA.2 has dominated the news cycle this week, we’re still learning more about—and seeing policy shifts due to—the original Omicron strain, BA.1. Here are a few major updates:
- Omicron is causing 100% of new COVID-19 cases in the U.S. According to the CDC’s latest update of its variant prevalence estimates, Omicron caused more than 99% of new COVID-19 cases in the country for the second week in a row: there was a slight increase from 99.4% of new cases in the week ending January 15 to 99.9% in the week ending January 22. The remaining 0.1% of cases are caused by Delta. The CDC is not yet distinguishing between BA.1 and BA.2 in its estimates, but will likely start doing so in the coming weeks.
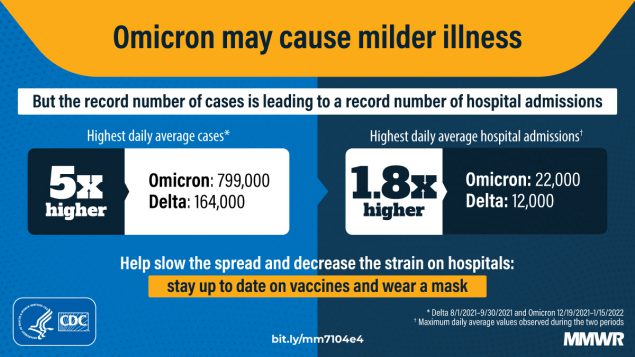
- ICU admissions and other indicators of severe COVID-19 symptoms were lower during the Omicron surge than during previous surges in the U.S. The CDC released another round of studies presenting Omicron’s impact on the healthcare system last week, including this report focusing on disease severity. CDC researchers analyzed data from three hospital surveillance systems and an additional large database; while the peak of new COVID-19 cases was five times higher during the Omicron surge than the Delta surge, they found, the peak of hospital admissions was only 1.8 times higher. And ICU admissions, the average stay length for hospitalized COVID-19 patients, and death rates were all lower during the Omicron surge than in the Delta and winter 2020-2021 surges.
- Pfizer and BioNTech have started a clinical trial of an Omicron-specific vaccine. Quickly after Omicron was identified, both Pfizer and Moderna started updating their vaccines for this variant and investigating whether such Omicron-specific shots would be necessary. Pfizer is now entering a new clinical trial with its Omicron-specific shot, along with its vaccine development partner BioNTech, according to Reuters. Trial participants will include about 1,400 people who received third doses from Pfizer’s current COVID-19 vaccine regimen three to six months earlier. Pfizer intends to study the Omicron-specific shot’s safety and effectiveness against this variant.
- The FDA recommends that U.S. facilities stop using monoclonal antibody treatments that don’t work well against Omicron. Speaking of Omicron-specific drugs: the Food and Drug Administration (FDA) announced this week that the agency is limiting use of two monoclonal antibody treatments, made by Regeneron and Eli Lilly. Both treatments, which boost patients’ immune systems by providing them with lab-made antibodies, worked well at reducing severe symptoms for past variants but have less of an impact on Omicron infections. As a result, healthcare providers should “ limit their use to only when the patient is likely to have been infected with or exposed to a variant that is susceptible to these treatments,” the FDA says. A third monoclonal antibody treatment, along with other drugs (including Pfizer’s hard-to-find pill), do work well against Omicron.
- Omicron has a short incubation period, so test timing is key. As I’ve written in the past, Omicron infections tend to move more quickly than infections with past variants: people can go from an exposure to symptoms within three days, rather than four or five. As a result, there’s less time to catch an infection before becoming contagious; but at the same time, rapid tests may give negative results in those early days of an infection, before patients have built up enough of a viral load for an antigen test to identify the infection. A recent New York Times article summarizes the science on how Omicron infections compare to past variants and provides recommendations on testing. For instance: “many experts now recommend taking a rapid test two to four days after a potential exposure,” and ideally testing twice about a day apart.
- Some experts are optimistic that we could see a COVID-19 lull after Omicron surges. Last week, I shared a STAT News article explaining that, thanks to high levels of population immunity, the U.S. might have “a bit of a break from the Covid roller coaster” after our Omicron surge ends in the coming weeks. Despite BA.2 concerns, Dr. Hans Henri Kluge, WHO’s regional director for Europe, shared a similarly optimistic view in a statement last week. “The pandemic is far from over, but I am hopeful we can end the emergency phase in 2022 and address other health threats that urgently require our attention,” he said.
Leave a comment