
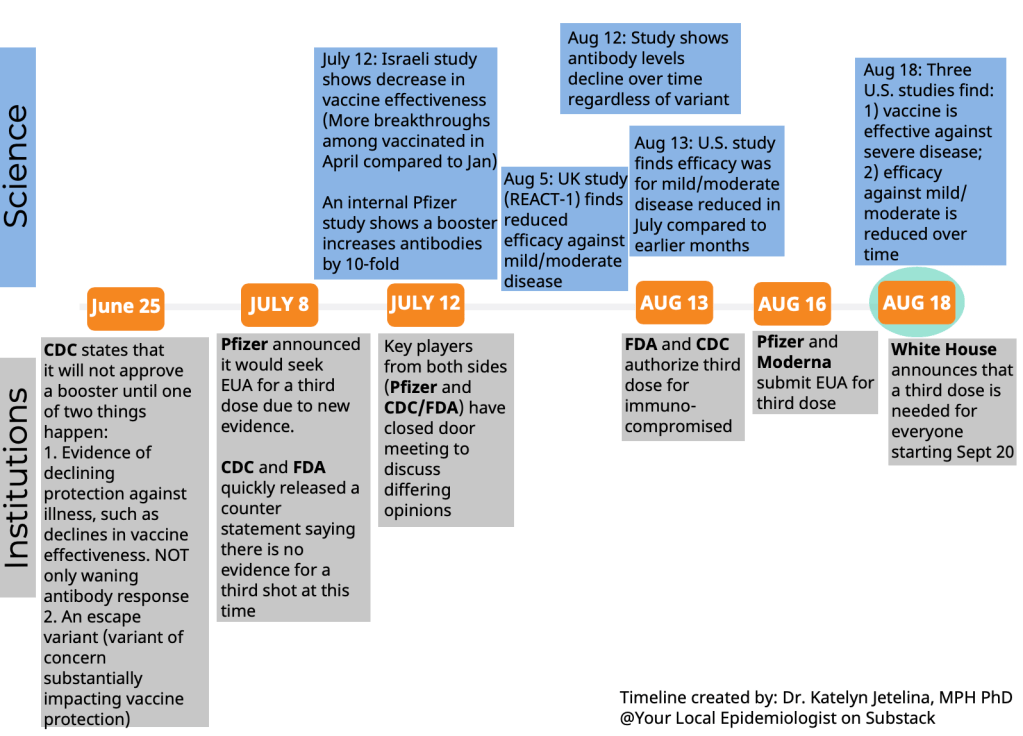
This week, the federal government announced that the U.S. intends to provide third vaccine doses to all Americans who received the Pfizer or Moderna vaccines. This booster shot distribution will start in September, with adults becoming eligible once they hit eight months after their second shot.
While the booster shot regimen still must be approved by the FDA and CDC, federal officials are making it sound like a pretty sure thing—President Biden himself announced the decision at a press conference on Wednesday. However, many epidemiologists, vaccine experts, global health experts, and other scientists have criticized the decision.
Here are three main criticisms I’ve seen in the past few days.
First: Scientific evidence is lacking. As the booster shot decision was announced on Wednesday, the CDC published three new studies that appear to show a decline in the Pfizer and Moderna vaccines’ ability to stave off symptomatic COVID-19 infection after several months. One of these reports, from a network of U.S. nursing homes, suggests that efficacy among nursing home residents fell to just 53% by June and July 2021, many months after this vulnerable population was vaccinated. The other two reports show similar declines, though the CDC found that vaccination remains effective against severe disease, hospitalization, and death.
The federal government—and others arguing in favor of booster shots—have also pointed to data from Israel, which appear to similarly demonstrate that the vaccines lose their effectiveness after several months. In Israel, where almost 80% of residents over age 12 are vaccinated, the majority of those hospitalized with COVID-19 are now fully vaccinated individuals.
But the act of interpreting these data is more complicated than it first appears. In a blog post at COVID-19 Data Science, biostatistics professor Jeffrey Morris explains that, when the majority of a population is vaccinated, vaccination numbers will go up in this population simply because they are the majority. But the risk remains far higher for the unvaccinated. Plus, Morris explains, stratifying hospitalization numbers by age reveals that older adults are more likely to have a severe COVID-19 case regardless of vaccination status, while younger adults are less likely to be vaccinated (and thus have a non-breakthrough case).
Simply put, the vaccines do still work well against severe COVID-19—you just need to be precise in calculating effectiveness. And yet, the U.S. government is saying that vaccine efficacy wanes so much, everyone’s going to need a third shot in the fall or early next year. This suggests that the federal government has more data that it is not sharing publicly, which leads us to the second criticism.
Second: Transparency is also lacking. Typically, when the government makes a decision about approving a new medical product, this decision follows a series of prescribed steps: data submission from the company behind the product, review by FDA scientists, FDA approval, followed by more review by other agencies (such as the CDC or the Centers for Medicare & Medicaid Services) as needed. Review meetings are typically open to the public, with data shared in advance of a decision. In the case of these booster shots, however, the president has announced a specific rollout plan before full scientific review has taken place.
As STAT’s Helen Branswell explains:
To many experts, including Baylor, the sequencing of the decisions being made is also out of whack. While U.S. health officials said booster shots could start being offered the week of Sept. 20, the Food and Drug Administration has not even ruled yet on Pfizer’s application for approval of a third shot; it was filed only Monday. Moderna hasn’t yet asked the agency to authorize a third shot at all.
Plus, remember that the CDC has not publicly shared any comprehensive data on breakthrough cases since the spring, before Delta became dominant.
The FDA and CDC will certainly still be reviewing the need for booster shots, but the experts cited in Branswell’s piece are skeptical that any decision other than, “Yes, go ahead” will be considered. I, for one, will be very curious to see how the discussions proceed—and what data get cited—at the FDA and CDC committee meetings.
Third: We need to vaccinate the world. As I’ve explained in the CDD before, getting vaccines to the low-income nations that have yet to start their rollouts is not just a humanitarian priority. It also protects us, here in the U.S., because the longer the coronavirus circulates, the more opportunities it has to mutate into increasingly-dangerous variants.
By moving to provide booster shots to everyone—not just the immunocompromised, the elderly, or the otherwise extra-vulnerable—the U.S. is likely delaying shots to other countries, prolonging the pandemic overall.
As Dr. Michael Ryan, emergencies chief at the World Health Organization, told reporters last week: “We’re planning to hand out extra life jackets to people who already have life jackets, while we’re leaving other people to drown without a single life jacket.”
More vaccine news
- Sources and updates, November 12Sources and updates for the week of November 12 include new vaccination data, a rapid test receiving FDA approval, treatment guidelines, and more.
- How is the CDC tracking the latest round of COVID-19 vaccines?Following the end of the federal public health emergency in May, the CDC has lost its authority to collect vaccination data from all state and local health agencies that keep immunization records. As a result, the CDC is no longer providing comprehensive vaccination numbers on its COVID-19 dashboards. But we still have some information about this year’s vaccination campaign, thanks to continued CDC efforts as well as reporting by other health agencies and research organizations.
- Sources and updates, October 8Sources and updates for the week of October 8 include new papers about booster shot uptake, at-home tests, and Long COVID symptoms.
- COVID source shout-out: Novavax’s booster is now availableThis week, the FDA authorized Novavax’s updated COVID-19 vaccine. Here’s why some people are excited to get Novavax’s vaccine this fall, as opposed to Pfizer’s or Moderna’s.
- COVID-19 vaccine issues: Stories from COVID-19 Data Dispatch readers across the U.S.Last week, I asked you, COVID-19 Data Dispatch readers, to send me your stories of challenges you experienced when trying to get this fall’s COVID-19 vaccines. I received 35 responses from readers across the country, demonstrating issues with insurance coverage, pharmacy logistics, and more.
Leave a comment