If the title of this week’s newsletter sounds ominous, that’s because this situation feels ominous. While many scientific experts have pushed back against President Trump’s claims that a vaccine for the novel coronavirus will be available this October, state public health agencies have been instructed to prepare for vaccine distribution starting in November or December.
Of course, the possibility of a COVID-19 vaccine before the end of 2020 is promising. The sooner healthcare workers and other essential workers can be inoculated, the better protected our healthcare system will be against future outbreaks. (And eventually, maybe, regular people like me will be able to attend concerts and fly out of the country again.) But considering the Center for Disease Control and Prevention (CDC)’s many missteps in both distributing and tracking COVID-19 tests this spring, I have a wealth of concerns about this federal agency’s ability to implement a national vaccination program.
I’m far from the only person thinking about this. The release of the CDC’s interim playbook for vaccine distribution this past Wednesday, along with President Trump’s public contradiction of the vaccination timeline described by CDC Director Dr. Robert Redfield, has sparked conversations on whether America could have a vaccine ready this fall and, if we do, what it would take to safely distribute this technology to the people who need it most.
In this issue, I will offer my takeaways on what the CDC’s playbook means for COVID-19 vaccination data, and a few key elements that I would like to see prioritized when public health agencies begin reporting on vaccinations.
Data takeaways from the CDC playbook
I’m not going to try to summarize the whole playbook here, because a. other journalists have already done a great job of this, and b. it would take up the whole newsletter. Here, I’m focusing specifically on what the CDC has told us about what vaccination data will be collected and how they will be reported.
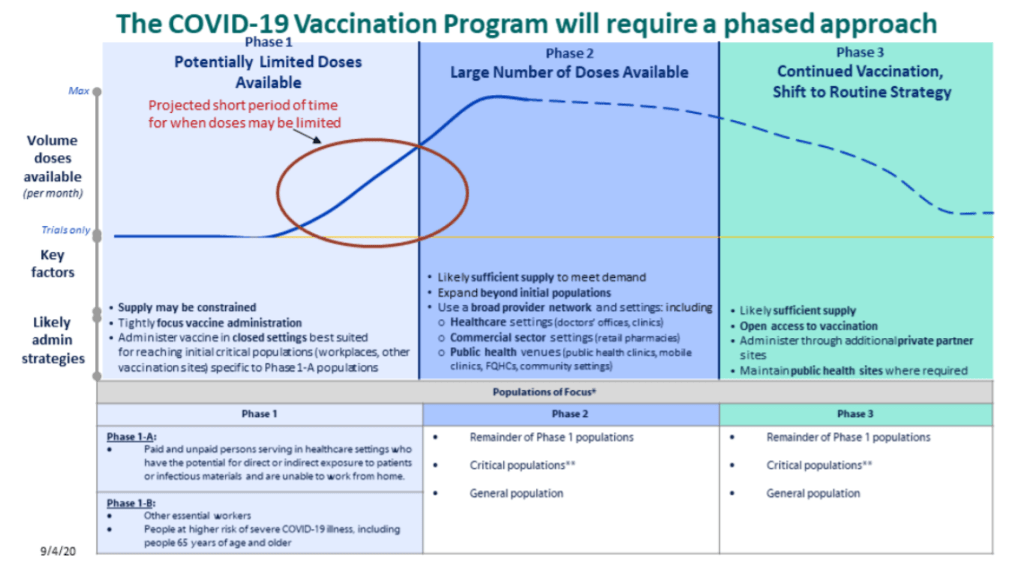
- We do not yet know which vaccines will be available, nor do we know vaccine volumes, timing, efficacy, or storage and handling requirements. It seems clear, however, that we should prepare for not just one COVID-19 vaccine but several, used in conjunction based on which vaccines are most readily available for a particular jurisdiction.
- Vaccination will occur in three stages (as pictured in the above graphic). First, limited doses will go to critical populations, such as healthcare workers, other essential workers, and the medically vulnerable. Second, more doses will go to the remainder of those critical populations, and vaccine availability will open up to the general public. Finally, anyone who wants a vaccine will be able to get one.
- “Critical populations,” as described by the CDC, basically include all groups who have been demonstrably more vulnerable to either contracting the virus or having a more severe case of COVID-19. The list ranges from healthcare workers, to racial and ethnic minorities, to long-term care facility residents, to people experiencing homelessness, to people who are under- or uninsured.
- The vaccine will be free to all recipients.
- Vaccine providers will include hospitals and pharmacies in the first phase, then should be expanded to clinics, workplaces, schools, community organizations, congregate living facilities, and more.
- Most of the COVID-19 vaccines that may come on the market will require two doses, separated by 21 or 28 days. For each recipient, both doses will need to come from the same manufacturer.
- Along with the vaccines themselves, the CDC will send supply kits to vaccine providers. The kits will include medical equipment, PPE, and—most notably for me—vaccination report cards. Medical staff are instructed to fill out these cards with a patient’s vaccine manufacturer, the date of their first dose, and the date by which they will need to complete their second dose. Staff and data systems should be prepared for patients to receive their two doses at two different locations.
- All vaccine providers will be required to report data to the CDC on a daily basis. When someone gets a vaccine, their information will need to be reported within 24 hours. Reports will go to the CDC’s Immunization Information System (IIS).
- The CDC has a long list of data fields that must be reported for every vaccination patient. You can read the full list here; I was glad to see that demographic fields such as race, ethnicity, and gender are included.
- The CDC has set up a data transferring system, called the Immunization Gateway (or IZ Gateway), which vaccine providers can use to send their daily data reports. Can is the operative word here; as long as providers are sending in daily reports, they are permitted to use other systems. (Context: the IZ Gateway is an all-new system which some local public health agencies see as redundant to their existing vaccine trackers, POLITICO reported earlier this week.)
- One resource linked in the playbook is a Data Quality Blueprint for immunization information systems. The blueprint prioritizes making vaccination information available, complete, valid, and timely.
- Vaccine providers are also required to report “adverse events following immunization” or poor patient outcomes that occur after a vaccine is administered. These outcomes can be directly connected to the vaccine or unrelated; tracking them helps vaccine manufacturers detect new adverse consequences and keep an eye on existing side effects. Vaccine providers are required to report these adverse events to the Vaccine Adverse Event Reporting System (VAERS), which, for some reason, is separate from the CDC’s primary IIS.
- Once COVID-19 vaccination begins, the CDC will report national vaccination data on a dashboard similar to the agency’s existing flu vaccination dashboard. According to the playbook, this dashboard will include estimates of the critical populations that will be prioritized for vaccination, locations of CDC-approved vaccine providers and their available supplies, and counts of how many vaccines have been administered.
I have to clarify, though: all of the guidelines set up in the CDC’s playbook reflect what should happen when vaccines are implemented. It remains to be seen whether already underfunded and understaffed public health agencies, hospitals, and health clinics will be able to store, handle, and distribute multiple vaccine types at once, to say nothing of adapting to another new federal data system.
My COVID-19 vaccination data wishlist
This second section is inspired by an opinion piece in STAT, in which physicians and public health experts Luciana Borio and Jesse L. Goodman outline three necessary conditions for effective vaccine distribution. They argue that confidence around FDA decisions, robust safety monitoring, and equitable distribution of vaccines are all key to getting this country inoculated.
The piece got me thinking: what would be my necessary conditions for effective vaccine data reporting? Here’s what I came up with; it amounts to a wishlist for available data at the federal, state, and local levels.
- Unified data definitions, established well before the first reported vaccination. Counts of people who are now inoculated should be reported in the same way in every state, county, and city. Counts of people who have received only one dose, as well as those who have experienced adverse effects, should similarly be reported consistently.
- No lumping of different vaccine types. Several vaccines will likely come on the market around the same time, and each one will have its own storage needs, procedures, and potential effects. While cumulative counts of how many people in a community have been vaccinated may be useful to track overall inoculation, it will be important for public health researchers and reporters to see exactly which vaccine types are being used where, and in what quantities.
- Demographic data. When the COVID Racial Data Tracker began collecting data in April, only 10 states were reporting some form of COVID-19 race and ethnicity data. North Dakota, the last state to begin reporting such data, did not do so until August. Now that the scale of COVID-19’s disproportionate impact on racial and ethnic minorities is well documented, such a delay in demographic data reporting for vaccination would be unacceptable. The CDC and local public health agencies will reportedly prioritize minority communities in vaccination, and they must report demographic data so that reporters like myself can hold them accountable to that priority.
- Vaccination counts for congregate facilities. The CDC specifically acknowledges that congregate facilities, from nursing homes to university dorms to homeless shelters, must be vaccination priorities. Just as we need demographic data to keep track of how minority communities are receiving vaccines, we need data on congregate facilities. And such data should be consistently reported from the first phase of vaccination, not added to dashboards sporadically and unevenly, as data on long-term care facilities have been reported so far.
- Easily accessible resources on where to get vaccinated. The CDC’s vaccination dashboard will reportedly include locations of CDC-approved vaccine providers. But will it include each provider’s open hours? Whether the provider requires advance appointments or allows walk-ins? Whether the provider has bilingual staff? How many vaccines are available daily or weekly at the site? To be complete, a database of vaccine providers needs to answer all the questions that an average American would have about the vaccination experience. And such a database needs to be publicized widely, from Dr. Redfield all the way to local mayors and school principals.
Leave a comment