
This week, the FDA’s vaccine advisory committee met to discuss fall booster shots, in anticipation of another COVID-19 surge next winter. The discussion demonstrated the U.S.’s continued failure to provide the data that are really needed to make these decisions.
I have written a lot about this topic in the past, so to avoid being too repetitive, I’ll link to a couple of past articles:
- The data problem underlying booster shot confusion (COVID-19 Data Dispatch, September 2021)
- The U.S. Is Relying On Other Countries’ Data To Make Its Booster Shot Decisions (FiveThirtyEight, November 2021)
- The U.S. still doesn’t have the data we need to make informed decisions on booster shots (COVID-19 Data Dispatch, April 2022)
But here’s the TL;DR: due to the fractured nature of America’s public health system, it’s difficult for researchers to connect data on different health metrics. For example, a state might have one database with vaccination records and another database with case records, and the databases might not easily link to answer questions about breakthrough cases.
Some state health departments have figured out how to make these links, but the process is not uniform. And the breakthrough case data we do have generally aren’t linked to information on variants, or demographic data, or outcomes like Long COVID.
The more specific the vaccine effectiveness question, the more complicated it becomes to answer. This is a bigger problem now as the FDA considers fall boosters, because the agency needs to determine the best vaccine candidate and identify priority populations for shots—while operating in a politcal climate where vaccine funding is less popular than it was a year ago.
Here are a few questions that the FDA is trying to answer, drawing from the STAT News meeting recap:
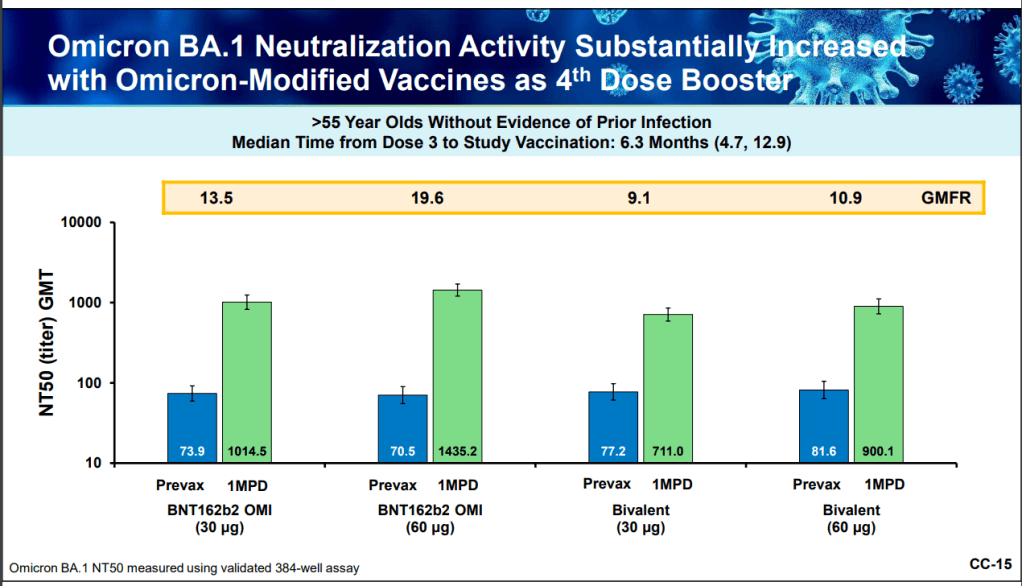
- Should the fall booster be a monovalent vaccine, meaning it only includes Omicron-specific genetic material? Or should it be bivalent, meaning it includes both Omicron and the original, Wuhan strain? Pfizer and Moderna presented different options; some experts say a bivalent vaccine may provide more long-term protection.
- Should the booster shot be specific to BA.4 and BA.5? The panel agreed that it should, as these strains are now dominant in the U.S., but there’s a timing trade-off as vaccine companies have yet to do clinical trials (or provide substantial data) for a subvariant-specific vaccine.
- Should the booster shot be another type of vaccine entirely? In addition to Pfizer and Moderna, the FDA panel also heard from Novavax. This company has developed a protein-based vaccine that hasn’t yet received FDA authorization, but panelists were impressed by its potential for long-term protection.
- How well do the vaccines provide non-antibody-based protection? As in past advisory committee meetings, the vaccine companies primarily presented data based on antibodies generated from their shots. Experts wanted to see more data about T cells and other aspects of immunity which are harder to measure, but may be more important in the long term.
- Who would most benefit from another booster? If the federal government isn’t able to buy enough shots for everyone, priortization will need to happen. Will Omicron-specific boosters be most useful for seniors, or for people with certain health conditions? These groups will likely get priority again, though we could still be collecting more data on how the vaccines fare for them.
Of course, despite the dearth of data and cautions from some members of the FDA advisory committee, the U.S. government seems to be going full-speed ahead with fall boosters. The Biden administration has placed a $3.2 billion order from Pfizer for 105 million doses of whichever Omicron-specific vaccine the FDA chooses to authorize.
Leave a comment