This past Friday, the Food and Drug Administration (FDA)’s vaccine advisory committee voted to recommend booster shots of the Pfizer-BioNTech vaccine for all Americans over age 65 and those who are particularly vulnerable to the virus, due to their health conditions and/or work environments. This was a notable recommendation because it went against the FDA’s ask: booster shots for everyone over the age of 16.
Let’s walk through the data behind this decision.
How is the current two-dose vaccine regimen faring against severe COVID-19 disease?
Before we get into any numbers, it’s important to remember the initial goal of the COVID-19 vaccines: protect people against severe disease, hospitalization, and death, basically reducing the coronavirus’ power to cause deadly harm.
On this front, all of the vaccines are performing well. Numerous papers cited during the advisory meeting, as well as the U.S.’s breakthrough case data, suggest that vaccination protects against severe COVID-19 disease for the vast majority of recipients. Among over 178 million people who had been fully vaccinated in the U.S. by mid-September, just 3,000 have died following a positive COVID-19 test. Those 3,000 deaths account for just about 1% of all COVID-19 deaths in the U.S. since January 2021.
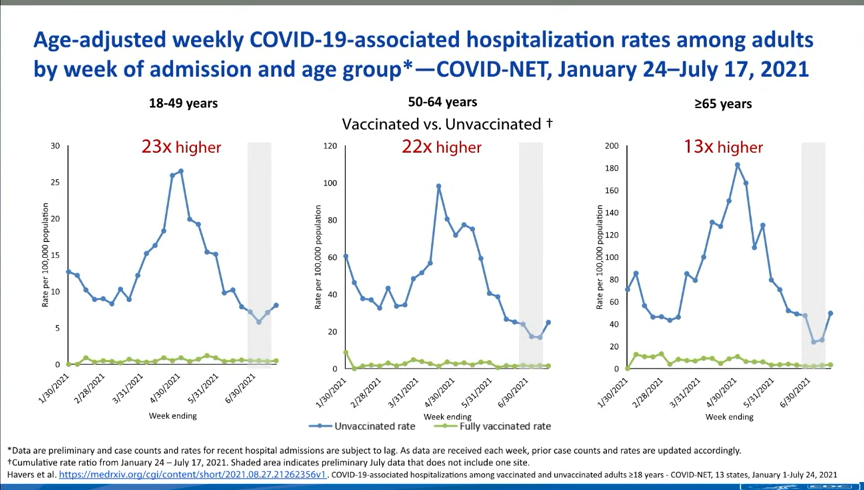
The numbers get a bit more complex, however, when you look at older adults and other vulnerable populations. Those who were more vulnerable to a severe COVID-19 case in the first place are also more vulnerable to having a severe breakthrough case, if they encounter the virus after vaccination. One chart, presented at the FDA meeting, provides a picture of this trend. From late January to mid-July, 2021, the hospitalization rate among younger adults (ages 18-49) was 23 times higher for the unvaccinated than for the vaccinated. For seniors (over age 65), however, the rate was 13 times higher for the unvaccinated.
How is that current regimen faring against coronavirus infection?
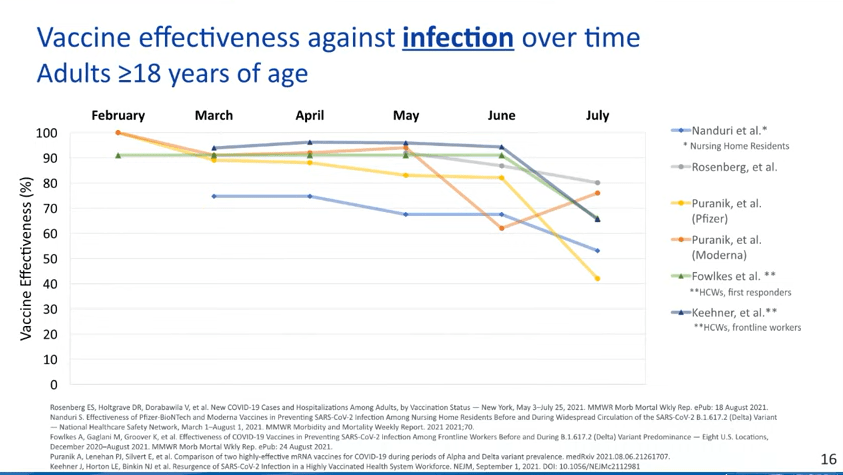
This is where we see a bigger drop in efficacy. Multiple studies point to the Pfizer and Moderna vaccines becoming less capable of protecting recipients against infection, over time; in other words, if you got your two shots in April 2021, you’re more likely to get a positive test result now, in September, than you were in May. (Though your case will likely be mild or asymptomatic!)

We can also see this in breakthrough case numbers when we look at all infections, as opposed to only those cases that lead to severe disease or death. This type of analysis is difficult to do in the U.S., as the CDC is only systematically tracking those severe cases, but we can see patterns in the data from local jurisdictions that are reporting their breakthrough cases more comprehensively.
For example, let’s look at Washington, DC, which reports breakthrough cases in extensive detail:

During the week of March 8, DC reported 14 breakthrough cases. The district reported about 800 cases overall that week, meaning that breakthroughs accounted for 2% of all cases.
During the week of August 23, however, the district reported almost 500 breakthrough cases. In that week, the district reported about 1,400 cases overall—meaning that breakthrough cases have jumped from 2% of all weekly DC cases to 35% of all weekly DC cases.
DC also reports a breakdown of breakthrough cases according to the time it’s been since residents were fully vaccinated. This reveals that most breakthroughs occur at least two months after an individual completed their dose series, with the highest number of breakthroughs in people who’d been vaccinated three to four months ago. We can assume that similar patterns are occurring elsewhere in the country.
It’s also worth noting that we don’t have a great sense of how well the vaccines protect against Long COVID—though data thus far suggest that post-vaccination Long COVID cases are much rarer than non-breakthrough cases.
Why are the vaccines appearing to lose their effectiveness?
This was a big point of discussion for the FDA advisory committee. Are the Pfizer and Moderna vaccines appearing to lose their ability to protect us against coronavirus infection because Delta has a special ability to evade the vaccines or because the vaccines become less effective over time?
One early-morning presenter at the FDA meeting, medical statistician Jonathan Sterne from the University of Bristol, dove into this issue. His presentation focused on confounders, a statistical term for an outside force that influences the question a researcher is trying to study. In the case of vaccine effectiveness, Sterne said, there are a lot of confounders; these include vaccine recipients’ ages, how long ago they were vaccinated, and when they were vaccinated (i.e. in which phase of the pandemic?).

Sterne and other British researchers have taken advantage of the U.K.’s extensive electronic health records to analyze how well the vaccines are working, attempting to take these confounders into consideration. Overall, he said, it’s very challenging to get trustworthy effectiveness numbers—though the U.K. has approved boosters for residents over age 50, so it’s clear that the country’s public health agency does see some need for the additional shots.
Sterne’s presentation, as did a presentation from Israeli public health officials, also underscored the need for the U.S. to collect more standardized data on breakthrough cases, among other things.
Why did the FDA advisory committee vote against booster shots for everyone, ages 16 and over?
When this advisory committee votes on a question regarding vaccines or another biological product, the committee is specifically asked to consider whether the benefits of the product outweigh the risks. In this case, do the benefits of widespread boosters outweigh the risks of potential side effects from those additional doses?
When it comes to those risks of potential side effects, the committee had strikingly little data to evaluate. Pfizer did conduct a clinical trial of booster shots, but it only included 306 participants—an incredibly small number, when compared to the massive trials of the vaccine’s original two-dose regimen. The trial didn’t include any participants under age 18 or over age 55, which some advisory committee members found problematic, as they were being asked to consider approval for all Americans over age 16.
Israel—which has now administered booster shots to over 2.8 million residents—provided some data on side effects, but their utility is limited. The country started giving boosters to older adults before moving to younger adults, limiting Israeli health officials’ ability to identify potential risk for myocarditis or other severe side effects that might be more common in the younger population.

While data from Israel do suggest that booster shots can bring down infection numbers in an overall population, the FDA advisory committee did not find that a sufficient argument to recommend boosters for all Americans. Not at this time, anyway.
Why did the committee vote to support boosters for seniors and other vulnerable populations?
The risks of booster shots may not be clear for younger adults, but the risks of a breakthrough COVID-19 case are clear for older adults and others with health conditions that make them more vulnerable to severe COVID-19 case. The committee’s vote to recommend boosters for vulnerable groups aligns with a growing scientific consensus: that the U.S. should protect seniors, nursing home residents, and others who are at higher risk for serious COVID-19 cases.
What happens next?
It’s important to underscore here that this booster shot recommendation came from a committee that advises the FDA, not from the FDA itself. The agency typically follows its committee’s recommendations, but it doesn’t have to. We can expect the FDA’s decision—approval of booster shots for vulnerable groups, for everyone over age 16, or something else—within a couple of days.
Next week, on Wednesday and Thursday, a CDC advisory committee is set to meet to further discuss booster shots. If both the FDA and CDC approve boosters, health departments across the country are prepared to begin administering them to eligible Americans; this will likely include seniors and other vulnerable adults who previously got two shots of the Pfizer vaccine.
What about everyone who got the Moderna or Johnson & Johnson vaccines?
Again, this decision focused on the Pfizer vaccine, so Moderna and J&J recipients will need to wait for more data and more deliberation. Moderna has formally applied to the FDA for authorization of its booster shot, so we may see a similar series of meetings about that vaccine in the coming weeks.
J&J vaccine recipients will likely experience a longer wait as researchers collect data on the effectiveness of this one-shot vaccine. CNET has a good explainer of the situation.
Also: If you’d like to read a more detailed breakdown of everything that happened at Friday’s advisory committee meeting, I highly recommend the STAT News liveblog by Helen Branswell and Matthew Herper, which I drew upon heavily in writing this post.
Leave a comment