You’ve probably heard about the Johnson & Johnson debacle by now. (Here’s an explainer on NYT’s The Daily and another one with more scientific background from Roxanne Khamsi at The Atlantic if you’re still confused.)
If you attended or read about the April 14 emergency ACIP (Advisory Committee on Immunization Practices) meeting discussing the pause, you probably heard “VAERS” a lot. VAERS stands for “Vaccine Adverse Event Reporting System.” It’s used as a “national early warning system to detect possible safety problems in U.S.-licensed vaccines,” it’s been around for much longer than the COVID-19 vaccines or even COVID-19, and it’s how regulators are examining the data about possible complications related to the Johnson & Johnson COVID-19 vaccine. And as the cherry on top, the data is open to the public through the WONDER search engine. So what’s there?
The search procedure is VERY customizable, and there’s even a video teaching you how to do it. You can narrow your search by symptoms, vaccine type, vaccine products, date that the event was reported, and more. For my cursory search, I, like a lot of people, was curious about the results for the Johnson & Johnson COVID-19 vaccine, so I just narrowed my search to “COVID-19 Vaccine Janssen.”
Here are my results, sorted by most common symptoms:
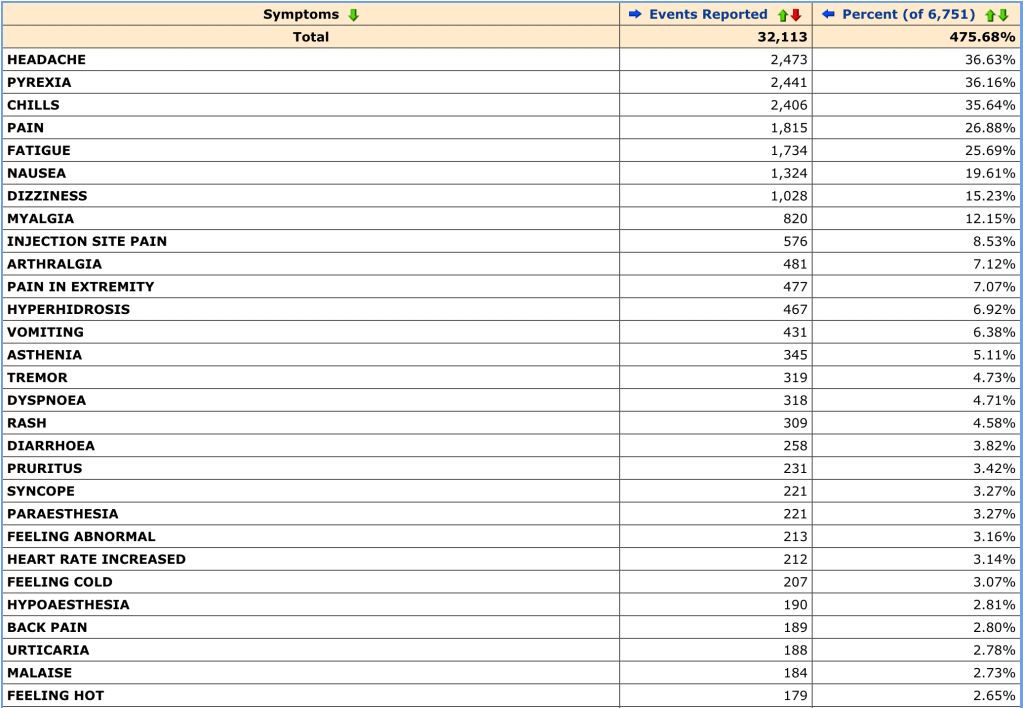
These are the most common results, but the list keeps going after that with rarer side effects as you go down. It quickly became clear that this is not a perfect system, with some slightly nonsensical reports and strange distinctions. I was certainly left wondering how “SARS-COV-2 test negative” ended up as an adverse effect. (Seems like that’s what you want? Either way, there were 59 of these reported. Good for those 59 people—or that one person with a very sore nose.)
Also notable were “Feeling abnormal” (213 reports), “Irritability” (10 reports) and my personal favorite, “No adverse event” (58 reports). It’s fairly obvious that not all of these adverse events were directly caused by the vaccine. Indeed, while the list can be fun to poke through and somewhat illuminating in what keeps popping up (headache, chills, pain, pyrexia/fever), finding the most common symptoms seems to be its main use.
J&J distribution is paused right now because there were 6 cases of cerebral venous sinus thrombosis (CVST) combined with low platelets in a group where that is not common, and symptoms manifested between 6 and 13 days after vaccination. However, you can’t see that from this dataset. It doesn’t even seem like this dataset is fully updated—right now, there’s only one case of CVST recorded. (It was last updated on April 10.) The dataset also doesn’t actually say when the person got vaccinated and when the event was reported—just frequency.
In the April 14 emergency ACIP meeting, Dr. Tom Shimabukuro of the CDC COVID-19 Vaccine Task Force cited the 6 reports as of April 12, so it looks like the public dataset hasn’t been updated while the people actually making decisions are working with updated data. It doesn’t mean that this dataset isn’t useful, it just means that one should act with caution before using it to draw any conclusions.
The CDC clearly agrees with me, as they make you check a box stating that you’ve read the disclaimers like “reports may include incomplete, inaccurate, coincidental and unverified information” and “the number of reports alone cannot be interpreted or used to reach conclusions about the existence, severity, frequency, or rates of problems associated with vaccines” before they show you the goods in WONDER.
There’s also a 17-page “VAERS Data Use Guide” which of course I found when I was almost done writing this up. Despite the limitations of the actual public dataset, I was extremely impressed by how much guidance that’s provided. I haven’t had my hand held this much while sifting through data in ages (I was very much on my own while going through the Global.Health database) and it really shows how much they want this data to be used with care. So I give serious props for solid and effective guidance on how to communicate this data—I just wish there was more data to communicate.
But WONDER isn’t the only way to sift through all the data. You can also download CSV files of every event reported (supposedly) back until 1990. That’d be a bit much for one post, but next week we might see what’s there.
More vaccine news
- Sources and updates, November 12Sources and updates for the week of November 12 include new vaccination data, a rapid test receiving FDA approval, treatment guidelines, and more.
- How is the CDC tracking the latest round of COVID-19 vaccines?Following the end of the federal public health emergency in May, the CDC has lost its authority to collect vaccination data from all state and local health agencies that keep immunization records. As a result, the CDC is no longer providing comprehensive vaccination numbers on its COVID-19 dashboards. But we still have some information about this year’s vaccination campaign, thanks to continued CDC efforts as well as reporting by other health agencies and research organizations.
- Sources and updates, October 8Sources and updates for the week of October 8 include new papers about booster shot uptake, at-home tests, and Long COVID symptoms.
- COVID source shout-out: Novavax’s booster is now availableThis week, the FDA authorized Novavax’s updated COVID-19 vaccine. Here’s why some people are excited to get Novavax’s vaccine this fall, as opposed to Pfizer’s or Moderna’s.
Leave a comment