It’s been one month since the Pfizer/BioNTech vaccine was authorized for use in the U.S. Since then, about 22 million Pfizer and Moderna doses have been distributed—and at least 6.7 million of those have actually made it into people’s arms, according to the CDC. (The CDC is not yet tracking second doses.)
Despite the federal government’s intense push to get vaccines through safety trials, that “last mile” step—from the Pfizer and Moderna factories to people’s arms—has been under-planned and underfunded. In the past month, we’ve been shocked by news stories ranging from a Wisconsin medical employee “intentionally removing” doses from a refrigerator to a local journalist in Florida individually helping over 150 seniors register for vaccination appointments.
State public health departments, already overwhelmed from ten months of running every other aspect of pandemic response, needed more money and resources to simultaneously coordinate millions of vaccinations and communicate their importance. The needed money didn’t come until this month, and recommendations from the federal government have left a lot of room for interpretation—leaving state and local health agencies scrambling.
And this first month was supposed to be the easy part! As The Atlantic’s Sarah Zhang explains, early U.S. vaccination efforts were aimed at easy-to-reach people: those in hospitals, nursing homes, and other long-term care facilities. In these locations, it’s easy to quickly identify the most vulnerable patients and get them registered for vaccination appointments. The next groups of eligible Americans will not be so easy to reach. Doctors’ offices, pharmacies, and many other businesses will need to figure out vaccine logistics while also ramping up campaigns to convince people to even get vaccinated in the first place.
(For a plain-language explanation of this issue that you can send to confused friends and relatives, I highly recommend the latest episode of the Sawbones podcast with Dr. Sydnee McElroy and Justin McElroy.)
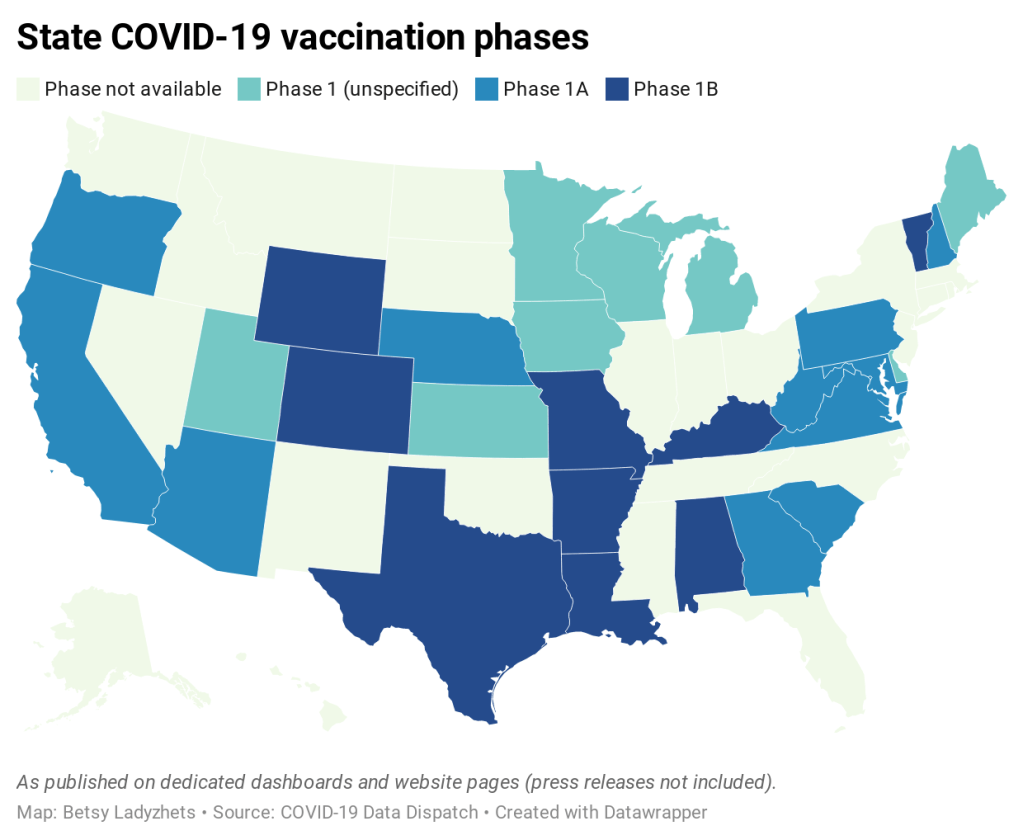
When I updated my vaccine data annotations yesterday, I added notes on how the vaccine rollout is progressing in each state. For the 38 states (and D.C.) now reporting vaccinations, you’ll find two new fields: the state’s vaccination phase (1A, 1B, etc.; 31 states are reporting this) and any prominently featured information on how residents can get vaccinated, such as a registration portal or contact information for local public health departments (at least 12 states are doing this).
Please note that, while most states do not yet have state-wide vaccine registration portals, many local public health departments are setting up such portals at the regional and county level. I highly recommend searching for your local public health agency to see what they have available. Also, New Mexico, which has a registration portal but no vaccine data dashboard, is not included in the annotations.
State data availability (as of Jan. 9)
- 39 jurisdictions are reporting some form of COVID-19 vaccination data on a dedicated page or dashboard
- 16 states are reporting race and ethnicity of vaccinated residents
- 20 states are reporting age of vaccinated residents
- 17 states are reporting gender or sex of vaccinated residents
- 20 states are reporting vaccinations by county or a similar local jurisdiction
- 31 states are reporting their vaccination phase (1A, 1B, etc.)
- 12 states are prominently featuring information on how residents can get vaccinated, such as a registration portal or contact information for local public health departments
More vaccine data news
- Jurisdictions with new vaccine dashboards or pages include: Arkansas, Arizona, California, Washington D.C., Kansas, Nebraska, and South Carolina.
- The CDC’s vaccination data are now available for download, via a table beneath the interactive dashboard. The agency updated its state-by-state data every weekday this past week—an improvement from the past two holiday weeks. A time series isn’t yet available, though.
- Bloomberg’s vaccine tracker now has time series for both individual states and several countries which have begun administering vaccines. The states currently leading the pack for vaccinations per capita in the U.S. are West Virginia, the Dakotas, and Maine.
- KFF has updated its COVID-19 Vaccination Monitor with polling data on vaccine hesitancy in rural America. Compared to urban and suburban residents, the foundation found, rural residents are significantly more hesitant. 31% of the rural residents sampled said they would “definitely get” a vaccine, compared to over 40% in other categories. Rural residents are also more likely to say they’re “not worried” that they or someone in their family will get sick with COVID-19.
- NPR’s Selena Simmons-Duffin and Pien Huang surveyed experts to determine several major ways the U.S. could “jump start its sluggish vaccine rollout.” These include: more money for state and local health departments, more vaccine types (hopefully some easier-to-transport brands), massive administration sites, more regular supplies from the federal government, and public awareness campaigns.
- The Trump administration is speeding up at least one thing: a plan to help pharmacies administer COVID-19 vaccines. According to POLITICO’s Rachel Roubein, almost 40,000 pharmacies are involved in the federal program, including those part of the Costco, Rite Aid, and Walmart chains. Pharmacies which are already used to administering flu vaccines each year—and already have huge patient databases—are strong candidates for the next phase of vaccine rollout.
- After some classic infighting from Governor Andrew Cuomo and Mayor Bill de Blasio, New York state is moving to Phase 1B—meaning seniors and essential workers will start to see vaccinations. However, as City Councilmember Mark Levine pointed out on Twitter, the city has: “One website for H+H sites, another for DOHMH sites, another for Costco. For community clinics, 7 have their own different websites, 4 require calling, and 1 is by email.” (I want to get vaccinated at Costco, personally, if the opportunity arises.)
- A Twitter thread from KFF Senior Vice President Jen Kates points out more of the methods states and counties are using to get residents signed up for vaccination appointments. They range from the expected online portals to SurveyMonkey and Eventbrite.
Related posts
- Sources and updates, November 12Sources and updates for the week of November 12 include new vaccination data, a rapid test receiving FDA approval, treatment guidelines, and more.
- How is the CDC tracking the latest round of COVID-19 vaccines?Following the end of the federal public health emergency in May, the CDC has lost its authority to collect vaccination data from all state and local health agencies that keep immunization records. As a result, the CDC is no longer providing comprehensive vaccination numbers on its COVID-19 dashboards. But we still have some information about this year’s vaccination campaign, thanks to continued CDC efforts as well as reporting by other health agencies and research organizations.
- Sources and updates, October 8Sources and updates for the week of October 8 include new papers about booster shot uptake, at-home tests, and Long COVID symptoms.
- COVID source shout-out: Novavax’s booster is now availableThis week, the FDA authorized Novavax’s updated COVID-19 vaccine. Here’s why some people are excited to get Novavax’s vaccine this fall, as opposed to Pfizer’s or Moderna’s.
Leave a comment